
FAM20C, Active (F18-11G)
FOR BULK ORDER REQUESTS PLEASE CONTACT US
Description :Recombinant human FAM20C (23-end) was expressed by baculovirus in Sf9 insect cells using an N-terminal GST tag.
Species :Human
Tag :GST tag
Expression System:Sf9 insect cells using baculovirus
Sequence :23-end
Genbank Number :BC040074
Specific Activity :Sample Kinase Activity Plot. For specific information on a given lot, see related technical data sheet.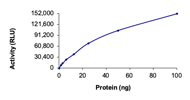
Purity :Sample Purity Data. For specific information on a given lot, see related technical data sheet. 
Storage, Stability and Shipping :Store product at –70oC. For optimal storage, aliquot target into smaller quantities after centrifugation and store at recommended temperature. For most favorable performance, avoid repeated handling and multiple freeze/thaw cycles.
Applications :Kinase Assay, Western Blot
Molecular Weight :93 kDa
Gene Aliases :GEF-CK, DMP4, G-CK, RNS
Scientific Background :FAM20C is Golgi casein kinase that phosphorylates secretory proteins within a Serx-Glu/pSer motif. It phosphorylates caseins and several other secretory proteins including AMTN, ENAM, SPP1 and the small integrin-binding ligand N-linked glycoproteins that are involved in biomineralization (1). FAM20C null mice develop hypophosphatemic rickets along with significant downregulation of osteoblast differentiation markers (2). Mutations in FAM20C gene are associated with Raine syndrome, an osteosclerotic bone dysplasia in humans (3). Additionally, FAM20C is implicated in other physiological processes, such as wound healing, regeneration, and cell migration (4).
References :
1. Cui, J. et al. (2015) A secretory kinase complex regulates extracellular protein phosphorylation. eLife. 4:e0612.
2. Wang X, et al: (2012) Inactivation of a novel FGF23 regulator, FAM20C, leads to hypophosphatemic rickets in mice. PLoS Genet. 8(5):e1002708.
3. Tagliabracci VS, et al: (2012) Secreted kinase phosphorylates extracellular proteins that regulate biomineralization. Science. 336(6085):1150–1153
4. Tagliabracci VS, et al: (2015) A single kinase generates the majority of the secreted phosphoproteome. Cell 161: 1619–1632.
Product Sheets (By Lot #) :
Research Areas :Cell Cycle, Inflammation, Metabolic Disorder, Ser/Thr Kinases
